Photoelectrochemical cells usually use indium tin oxide as a transparent conductor, but this material is relatively costly, mechanically fragile, and can lose its transmittance and conductivity at the potentials used for water splitting. In addition, if one coats the indium tin oxide with a uniform catalyst film, the catalyst can block light from reaching the dye or photovoltaic component of the photoelectrochemical cell.The challenge was to create a stable, earth-abundant, transparent, and electrically conductive catalyst for photoelectrochemical cells. We hypothesized we could create such a catalyst with a film made from core-shell nanowires. We showed that Cu-based nanowires (Cu, Cu-Ni, and Cu-Pt) could serve as a mechanically flexible, earth-abundant alternative to ITO for use in water splitting. For example, Cu-Ni nanowire networks could provide the same water oxidation performance as thin film catalysts on ITO, while transmitting up to 6.7 times more light (see Figure 1 and Angew. Chem. Int. Ed., 2013). Cu–Pt core–shell nanowire networks exhibited mass activities up to eight times higher than carbon-supported Pt nanoparticles for the hydrogen evolution reaction. The greater transmittance, mechancial flexibility, and lower materials cost of nanowire network catalysts relative to thin film catalysts deposited on ITO opens up new possibilities to engineer more efficient, mechanically robust, and affordable light-harvesting architectures.
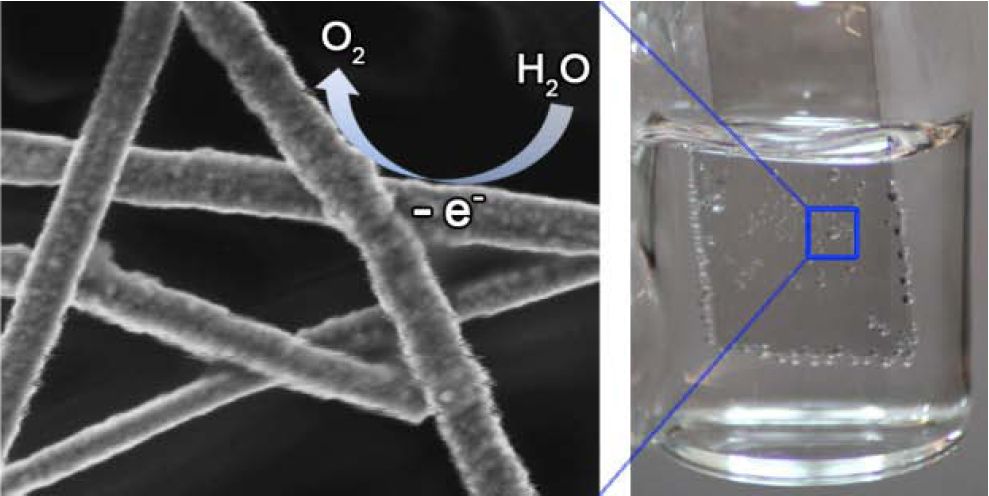
Figure 1. A transparent water oxidation catalyst based on nickel-coated copper nanowires.See the paper for more details
Related Publications:
Chen, Z.; Rathmell, A. R.; Ye, S.; Wilson, A. R.; Wiley, B. J. Optically Transparent Water Oxidation Catalysts Based on Copper Nanowires. Angew. Chem. Int. Ed. 2013, 52, 13708-13711.
Chen, Z.; Ye, S.; Wil¬son, A.R.; Ha, Y.-C.; Wiley, B.J. Optically Transparent Hydrogen Evolution Catalysts made from Networks of Copper-Platinum Core-Shell Nanowires. Energy Environ. Sci., 2014, 7, 1461-1467.
Du, J.; Chen, Z.; Ye, S.; Wiley, B.J.; Meyer, T.J. Copper as a Robust and Transparent Electrocatalyst for Water Oxidation. Angew. Chem. Int. Ed., 2015, 54, 2073-2078.